Share this link via
Or copy link




Article filters
Search Papers:
byMagda Rocío Gómez Marroquín*, Sandra L Carmona, Diana Burbano David, Andrea del Pilar Villarreal, Mauricio Soto Suárez, Adriana González Almario
Received: 15/June/2025 – Published: 29/April/2026
DOI: https://doi.org/10.18781/R.MEX.FIT.2401-3
Abstract Background/Objective. Bioactive substances such as phosphites and silicon present a promising alternative for management of vascular wilt of tomato, due to their potential to inhibit pathogen growth and to induce plant defense mechanisms. The objective of this study was to evaluate the effect of three phosphite sources and a source of silicon on isolate Fol59 of Fusarium oxysporum f. sp. lycopersici race 2 via in vitro and in planta assays.
Materials and Methods. In in vitro conditions, percentage of radial growth inhibition (PRGI) of isolate Fol59 was determined in PDA medium supplemented with each bioactive substance in concentrations between 10 and 20 000 ppm following a completely randomized design (4 × 9) with four treatments and nine different concentrations; five technical replicates and three biological replicates. In in planta conditions, the area under the disease progress curve (AUDPC) and efficacy percentage were evaluated with a completely randomized design (4 × 2) with four treatments and two different concentrations under four experimental conditions: control, inoculated control, treatments without inoculation and inoculated treatments. 20 experimental units and three biological replicates were used per treatment.
Results. After seven days, the bioactive substances reduced radial growth of the fungus between 70 and 100% with significant differences among the concentrations. The largest inhibition was registered with silicon at 10 000 ppm (100%) followed by calcium phosphite at 2 000 ppm (99%). Fourteen days post inoculation, in in planta assays, the control showed 100% incidence and 98% severity. However, the potassium phosphite treatment (KPhi1) at 2 000 ppm reduced severity to 56%, which represents a 42% efficacy against the control.
Conclusion. Phosphites and silicon showed inhibitory effect on mycelial growth of the fungus under in vitro conditions. In in planta assays, potassium phosphite at 2 000 ppm was the most effective treatment, reducing the severity of vascular wilt of tomato caused by Fol59.
byLeandris Argentel Martínez, Ofelda Peñuelas Rubio*, Francisco Cervantes Ortíz, Joe Luis Arias Moscoso, Francisco Cadena Cadena, Pamela Romo Rodríguez, Lorenzo Pérez López, Rosario Alicia Fierro Coronado
Received: 15/January/2026 – Published: 29/April/2026
DOI: https://doi.org/10.18781/R.MEX.FIT.2601-2
Abstract Background/Objective. The genus Fusarium comprises some of the most important phytopathogenic fungi affecting maize crops. Fusarium cause diseases such as root, stalk, and ear rot, which negatively impact the crop's agricultural productivity. In the search for agrobiotechnological alternatives for controlling this fungus, this study was aimed to evaluate the antagonistic potential of Parkinsonia aculeata rhizobacteria against Fusarium spp. strains associated with native maize varieties from the Bajío region of Mexico.
Experimental development. Two strains of Fusarium spp. (MC-03 and MC-05), isolated from roots of native maize varieties from the Bajío region of Mexico, exhibiting Fusarium wilt symptoms, were used. These fungi were tested in vitro against nine rhizobacteria of P. aculeata: Enterobacter cloacae (BA1), Priestia megaterium (BA4 and BA-7B), Sinomonas halotolerans (BA10-B), Staphylococcus warneri (BP5), P. endophytica (BP6), Bacillus subtilis (TP1 and TP2), and S. hominis (TM6). Microscopic and macroscopic characterization of the fungal strains and biochemical analysis of the rhizobacteria were performed. Radial growth inhibition of the fungal isolates was determined by triplicate dual fungus-rhizobacteria confrontations. A completely randomized design was used, analyzing the data obtained in the STATISTICA software using an ANOVA based on a linear model of fixed effects and a mean comparison test by DMS (p>0.05).
Results. With the exception of P. endophytica (BP6), all rhizobacteria exhibited enzymatic activity related to fungal antagonism mechanisms. B. subtilis (TP1) produced glucanases, lipases, and proteases. Microscopic and macroscopic characterization of the fungal strains indicated that they belong to Fusarium spp. In the rhizobacteria-MC-03 confrontation, the B. subtilis bacterial strains (TP1 and TP2) were statistically similar and achieved the greatest inhibition of mycelial growth (23%). With the fungal strain MC-05, P. endophytica (BP6) and S. hominis (TM6) were statistically superior to the other rhizobacteria in inhibiting mycelial growth (17%), followed by the two B. subtilis strains (TP1 and TP2), which inhibited 10% and 17%, respectively.
Conclusion. There was a significant variability in the mycelial growth response of the fungi to rhizobacteria. B. subtilis (TP1 and TP2), S. hominis (TM6), and P. endophytica (BP6) exhibited an antagonistic effect, inhibiting the mycelial growth of Fusarium spp. strains by up to 23% compared to the absolute and commercial controls. This study establishes the preliminary scientific basis for obtaining a biofungicide with specific inhibitory capabilities against the fungi studied.
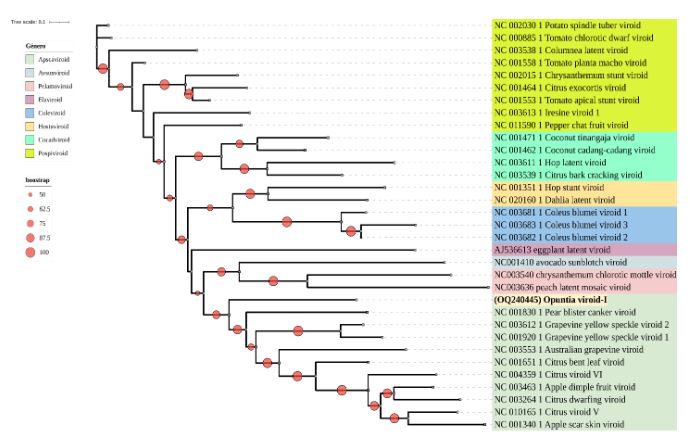
Detection of a tentative alphanucleorhabdovirus infecting Carica papaya in Costa Rica
byLaura Garita Salazar, William Villalobos Muller, Mauricio Montero Astúa, Antonio Bogantes Arias, Teresita Coto Morales, Izayana Sandoval Carvajal, Lisela Moreira Carmona*
Received: 11/February/2026 – Published: 29/April/2026
DOI: https://doi.org/10.18781/R.MEX.FIT.2602-3
Abstract Background/Objective. Foliar chlorosis, short internodes, and curved petioles with purple streaks were observed in papaya (Carica papaya) crops (North and Atlantic regions) in Costa Rica since 2014. Identification of a putative plant virus associated with these symptoms was the aim of this research.
Experimental development. Plant material was tested by ELISA (four plant viruses and potyviruses group), transmission electron microscopy (TEM), RT-PCR (degenerate primers for plant viruses), sequencing and phylogenetics.
Results. All ELISA tests resulted negative. Bullet-shaped particles inside nuclei, and reticulum endoplasmic were only observed by TEM in symptomatic plants. Amplicons of 900 bp were consistently obtained from symptomatic samples using degenerate primers for plant rhabdoviruses. Nucleotide sequences showed 95.6 and 96.8% similarity to a putative papaya alphanucleorhabdovirus (Alphanucleorhabdovirus, Rhabdoviridae).
Conclusion. This is the first report of a putative alphanucleorhabdovirus associated with symptomatic papaya plants showing streaked petiole ("pecíolo rayado") disease in Costa Rica, but Koch's postulates must be fulfilled and vector identified.
byCynthia Ford Villalón, David Schneider, Idalia Montesinos Solano, Jimena Carrillo Tripp*
Received: 13/December/2025 – Published: 23/April/2026
DOI: https://doi.org/10.18781/R.MEX.FIT.2512-4
Abstract Background/Objective. Grapevine red blotch virus, GRBV (Grablovirus vitis) affects grapevine plants (Vitis vinifera) causing significant economic losses in vineyards. In Mexico, where GRBV has been reported in wine-producing regions such as Baja California, information on potential GRBV vectors remains limited. Although the three-cornered alfalfa hopper (Spissistilus festinus) is a confirmed vector in the USA, the role of other membracids in vineyards around the globe is still unclear. Recently, the Nearctic treehopper (Tortistilus wickhami) has been reported in Baja California, prompting this study to evaluate its potential for GRBV acquisition and transmission.
Experimental development. Adult Nearctic treehoppers were collected from February to November 2023 in 20 vineyards in Valle de Guadalupe, Baja California. A total of 30 individuals were screened to detect GRBV by real-time PCR. In May 2024, 17 additional individuals were collected and used in transmission assays, with an acquisition access period of up to 4 days on GRBV-infected grapevine cv. Cabernet Sauvignon leaves, followed by an inoculation access period of up to 9 days on virus-free leaves. Both insects and recipient leaves were tested for GRBV by real-time PCR.
Results. GRBV was detected in 6.7% (2/30) of the individuals collected in 2023, with positive insects only originating from vineyards confirmed as GRBV-positive. In the transmission assays, 53% (9/17) of the insects acquired viral particles after feeding on infected leaves; however, none of the recipient leaves showed detectable infection.
Conclusion. This study demonstrates that the Nearctic treehopper, an insect phylogenetically close to, and morphologically resemblant of the three-cornered alfalfa treehopper (a confirmed GRBV vector), was able to acquire the virus in the field and in laboratory settings, but no transmission to recipient plants was proved under the tested experimental conditions in the laboratory.
bySebastian Iglesias Osores*, Luis A Alvarez
Received: 08/December/2025 – Published: 23/April/2026
DOI: https://doi.org/10.18781/R.MEX.FIT.2512-2
Abstract Background/Objective. Blueberry powdery mildew, associated with Erysiphe vaccinii, is a foliar disease that may cause defoliation and affect crop performance. In Peru, information on the foliar use of peracetic acid for its field management is limited. The objective of this study was to evaluate the effect of foliar applications of 15% peracetic acid on powdery mildew incidence in blueberry under field conditions in Ica, Peru.
Experimental development. The trial was conducted in a commercial field of blueberry cv. Biloxi in Ica, Peru, using a randomized complete block design with five treatments: an untreated control and 15% peracetic acid at 2, 4, 6, and 8 mL L-1, with four replicates. Treatments were applied as foliar sprays twice, seven days apart, at a spray volume of 600 L ha-1. The response variable was the incidence of leaves with powdery mildew symptoms (under natural infection), assessed on seven dates from the first application onward. The presence of phytotoxicity symptoms was also visually recorded.
Results. Before the first application, no differences were detected among treatments. After the first application, peracetic acid reduced powdery mildew incidence compared with the control, with initial efficacy of up to 67.7%. After the second application, the 4, 6, and 8 mL L-1 doses showed the best response, with maximum efficacies of 69.3, 78.0, and 79.7%, respectively. The 2 mL L-1 dose showed a lower effect. No visible phytotoxicity symptoms were observed at any of the evaluated doses.
Conclusion. Under the conditions of this trial, 15% peracetic acid reduced foliar powdery mildew incidence in blueberry, with the best response at 4, 6, and 8 mL L-1 (69.3, 78.0, and 79.7%). These results indicate a favorable effect of the product under field conditions, although further evaluation in additional locations or growing seasons is needed for validation.
byMiguel Salvador Figueroa, José Fernando Gómez López, Miguel Salvador Adriano, María de Lourdes Adriano Anaya, Isidro Ovando Medina, Benjamín Moreno Castillo*
Received: 26/August/2025 – Published: 11/March/2026
DOI: https://doi.org/10.18781/R.MEX.FIT.2408-4
Abstract Background/Objective. The production and quality of tomato (Solanum lycopersicum) crops are severely reduced by the fungus Fusarium oxysporum, the causal agent of fusarium wilt. Chemical fungicides are conventionally applied, although in cases of severe infection, the entire harvest is lost. Favorable environmental conditions increase the incidence, infection rate, and spread of this pathogen. Biological control is a useful strategy for combating this type of pathogen. The objective of this research was to determine the incidence of fusarium wilt (Fusarium oxysporum) in field-grown tomato plants treated with Trichoderma asperellum Jc01 and Bacillus subtilis ANT01.
Experimental development. Trichoderma asperellum Jc01 and Bacillus subtilis ANT01 either alone or in combination, were weekly applied to the drip zone of tomato plants (Solanum lycopersicum). Disease incidence was periodically sampled in both treated and untreated plants to monitor its progression over 15 weeks. Additionally, the number of flowers and fruits produced on the experimental plants was recorded.
Results. At the end of the field trial, plants treated with B. subtilis ANT01 showed 60% less incidence compared to the control and a total of 47.9 fruits produced per plant, while plants treated with Trichoderma asperellum Jc01 or the combination of both microorganisms showed 16 and 28% less incidence than the control, and 18.0 and 27.3 fruits produced per plant, respectively.
Conclusion. The results show at least partial evidence of the potential of the ANT01 strain as a biocontrol agent of F. oxysporum in tomato plants.
byGuillermo Ascencio Luciano, Nicolás Maldonado Moreno, Reinaldo Méndez Aguilar, Moisés Felipe Victoriano*
Received: 21/August/2024 – Published: 06/March/2026
DOI: https://doi.org/10.18781/R.MEX.FIT.2408-00
Abstract Background/Objective. The soybean crop is affected by various diseases caused by fungi, which can severely reduce yields, in some cases up to 100%. The objective of this study was to identify the fungus associated with leaf spot in soybean and to evaluate the influence of soil moisture on the incidence and severity of the disease in plant and leaf in 30 soybean genotypes.
Materials and Methods. Three soybean leaves with leaf spot symptoms were collected per genotype for morphological identification of the fungus using taxonomic keys. Two moisture conditions (irrigated and rainfed) were evaluated. Plant incidence was estimated from 10 randomly selected plants; during the same period, foliar incidence and severity were evaluated. For foliar incidence, healthy and diseased leaves were counted from 10 plants per genotype. Subsequently, from 10 plants per genotype, one leaf in the fifth position from top to bottom was selected. The leaves were photographed, and the damaged area and total leaf area were calculated using the ImageJ software, thereby determining leaf severity.
Results. Alternaria alternata was identified as associated with leaf spot. In the rainfed environment, leaf incidence by A. alternata ranged from 4.4 to 50.55%; for severity, the values ranged between 0.3 and 17.4%. In the irrigated environment, the incidence varied from 7 to 50.9%; the severity ranged from 0 to 24.7%.
Conclusion. A. alternata was associated with leaf spot in soybean, which is little studied in Mexico, and currently its effects on this crop are unknown; therefore, it is of scientific interest to continue working on research projects with this species.
Development and validation of a diagrammatic scale to assess the severity of cucumber downy mildew
byJuan Carlos Gonzalez Acevedo, Santiago Domínguez Monge*, Dagoberto Guillen Sánchez, Víctor López Martínez, Porfirio Juárez López, Irán Alia Tejacal, Julio David Mendoza García
Received: 05/January/2026 – Published: 06/March/2026
DOI: https://doi.org/10.18781/R.MEX.FIT.2601-1
Abstract Background/Objective. Cucumber downy mildew (Cucumis sativus), caused by Pseudoperonospora cubensis, is one of the major foliar diseases affecting cucumber production in Morelos, Mexico. Due to the lack of standardized and validated methods for quantifying disease severity, the objective of this study was to develop and validate a diagrammatic scale for its assessment.
Experimental development. A diagrammatic scale composed of eight severity levels (2, 4, 8, 16, 24, 32, 40, and 47% affected leaf area) was developed using naturally infected leaves whose actual severity was determined through digital image analysis with Assess®️ software. Validation of the scale was conducted with the participation of 20 raters, who visually estimated the severity of 50 leaves with different damage levels in three successive evaluations: one without the aid of the scale (WO) and two with the aid of the scale conducted two (A1) and four weeks (A2) after the initial evaluation.
Results. The use of the diagrammatic scale significantly increased the accuracy of severity estimates, with Pearson´s correlation coefficients of r = 0.91 and r = 0.93 in A1 and A2, respectively, compared to the evaluation without the scale (r = 0.34). Likewise, generalized bias was lower when the scale was used (Cb > 0.9), which was reflected in greater overall agreement between visual estimates and real severity values (ρc > 0.9).
Conclusion. The developed diagrammatic scale is a reliable, accurate, and reproducible tool for estimating cucumber downy mildew severity under field conditions.
Strategies to reduce losses from Fusarium graminearum head blight in bread wheat
byVíctor Manuel Arzola Mora, Héctor Eduardo Villaseñor Mir, Salvador Carranza González*, Carlos Sánchez Abarca, René Hortelano Santa Rosa, Julio Huerta Espino, María Elsa Rodríguez Contreras, Pawan Kumar Singh, Xinyao He
Received: 14/October/2025 – Published: 02/March/2026
DOI: https://doi.org/10.18781/R.MEX.FIT.2510-3
Abstract Background/Objective. Head blight (fusarium), caused by Fusarium graminearum in bread wheat, reduces crop yield and contaminates the grain with mycotoxins such as deoxynivalenol (DON), which affects the quality and safety of the product. Its severity increases in warm and humid areas during flowering, which highlights the need for integrated management strategies. The present study aimed to evaluate the interaction between three wheat genotypes (Heilo, Falcin and Ocoroni F86) and three fungicides Tilt® 250 EC (25% propiconazole), Nativo® 300 SC [trifloxistrobin (20%) + tebuconazole (10%)); and Folicur® 250 EW (tebuconazole (25%)], in order to reduce the severity of F. graminearum and improve yield under field conditions during two production cycles.
Experimental development. The experiment was carried out at the INIFAP Valley of Mexico Experimental Field during the spring-summer 2022 and 2023 cycles, a split plot design with three replications was used. Agronomic variables such as days to flowering (DF), days to maturity (DM), plant height (ALT), grain yield (REND) and severity of F. graminearum in the head (FEsp) were recorded. Statistical analysis was carried out using ANOVA, Duncan´s test (p<0.05) and Pearson´s correlations.
Results. The Heilo variety, when propiconazole (25%) or trifloxistrobin (20%) + tebuconazole (10%) was applied, showed the highest yield (>4 000 kg ha⁻¹) and the lowest severity of fusarium head scab (FEsp ≤1.5). In contrast, Ocoroni F86 was susceptible, even under chemical treatment. Climatic conditions had a significant effect on the severity of the disease.
Conclusion. The combines use of varieties with genetic resistance and triazole fungicides is a feasible agronomic strategy to mitigate fusarium head scab. It is recommended to validate these results in different environments to expand their applicability.
byEdith Luna Martínez, Sergio Aranda Ocampo*, Dimas Mejía Sánchez, Candelario Ortega Acosta, Manuel Livera Muñoz, Laura Delia Ortega Arenas, Antonio Mora Aguilera, Eligio Pérez Sosa, Daniel Telìz Ortiz
Received: 08/December/2025 – Published: 25/February/2026
DOI: https://doi.org/10.18781/R.MEX.FIT.2512-3
Abstract Background/Objective. The cultivation of pitahaya (Selenicereus undatus) has expanded in recent years in Mexico. In 2023, symptoms of stem rot were observed in commercial pitahaya orchards in the state of Puebla. The objective was to characterize and identify the causal agent of stem rot, evaluate the pathogenicity of the causal agent in different pitahaya species as well as other plant species, and assess its in vitro sensitivity to commercial bactericide formulations.
Materials and Methods. Ten rotten stems from Huitziltepec, Puebla, were analyzed. Tissue samples from each stem were disinfected and macerated in 500 μL of sterile distilled water; from there, 100 μL were seeded in Wilbrink's and King's B culture media. From the bacterial growth, strain CPHU23 was biochemically characterized and identified by multilocus sequence analysis (MLSA) and phylogenetic analysis of the rpoB, gyrB, leuS, and fusA genes. The pathogenicity of CPHU23 was evaluated by infiltration of 3 × 10⁸ CFU mL⁻¹ in five species of the genus Selenicereus: S. ocamponis, S. purpusi, Selenicereus sp. “Golden,” Selenicereus sp. “Solferina” and S. undatus, as well as in Agave cupreata, A. angustifolia, Aloe vera, Allium cepa, Solanum lycopersicum, and Capsicum annuum. In vitro sensitivity was determined by the disk diffusion method with 10 commercial bactericide formulations.
Results. Three bacterial morphotypes were isolated from the 10 stems. The CPHU23 morphotype was the most common, with yellow, round colonies with smooth edges and a mucoid appearance. Biochemical characterization of the CPHU23 strain showed 93% similarity to Pantoea vagans. MLSA analysis phylogenetically grouped the CPHU23 isolate within the Pantoea vagans clade, phylogenetically related to the type strains of P. vagans LMG 24199, YI-1, and MA I6050 with bootstrap support greater than 85%. Inoculation with P. vagans CPHU23 caused stem rot in the five species of the genus Selenicereus, in leaves of A. cupreata and A. vera, but not in bulbs of A. cepa and fruits of S. lycopersicum and C. annuum. Pantoea vagans CPHU23 was sensitive in vitro to copper oxychloride, gentamicin sulfate, oxytetracycline hydrochloride, and streptomycin, but resistant to kasugamycin.
Conclusion. Pantoea vagans is the causal agent of stem rot in pitahaya in Puebla. All five pitahaya varieties are susceptible to stem rot. Agave cupreata and Aloe vera could be potential hosts for P. vagans. The use of copper oxychloride may be an efficient management strategy for P. vagans in pitahaya cultivation in Mexico.